 One way to do this is to look for whether the machining shop is ISO 13485:2016 certified. 
How can you ascertain the validity of a company’s QMS? What sets a quality management system (QMS) above the rest for medical device manufacturing and machining? Safety, sanitation and product integrity are, without a doubt, crucial for any plastics machining company working with materials for medical use.  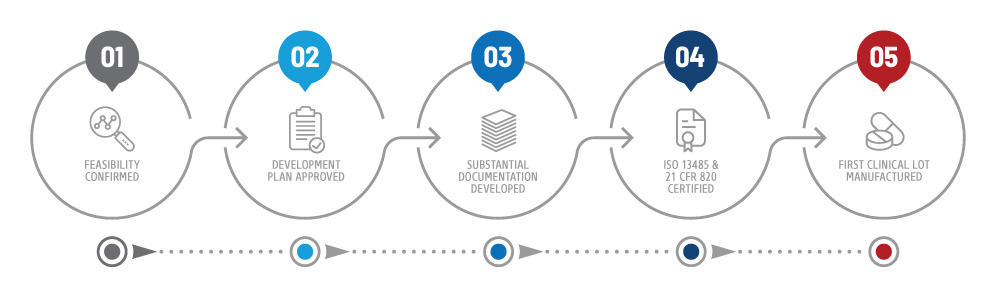
A discussion with the plastics pros at AIP on how the ISO 13485:2016 standard improves quality assurance for medical machined plastics. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
